Robert Harrod
Professor
Biological Sciences
| Office Location |
DLSB 334 |
| Phone |
214-768-3864 |
Education
Ph.D., University of MarylandPostdoc, National Institutes of Health and USUHS/NMC
Tel: 214-768-1965
Research Interests
Molecular Mechanisms of Viral Carcinogenesis: HTLVs and High-Risk Subtype HPVs
My laboratory is studying how certain transforming viruses cause cancers in humans. It is estimated approximately 15-20% of human cancers are caused by oncogenic viruses, yet the molecular etiology by which infectious agents deregulate host cellular growth/proliferative pathways to promote the establishment and progression of neoplastic disease is not completely understood. Our research is primarily focused toward advancing our understanding of the molecular biological and biochemical events underlying carcinogenesis by the human T-cell leukemia virus type-1 (HTLV-1) and high-risk subtype human papillomaviruses (HPVs).
The HTLV-1 is a complex oncoretrovirus that infects and transforms CD4+ T-lymphocytes and causes adult T-cell leukemia/lymphoma (ATLL) –an aggressive and often-fatal hematological malignancy that is highly resistant to most anticancer treatments. At present, there are 10-20 million HTLV-1-infected individuals worldwide, with most clustered in the tropical endemic regions of Southeast Asia (i.e., Japan, Taiwan, Malaysia, and the Philippines), the Middle East, Northern and Central Africa, Central and South America, Australo-Melanesia, and certain islands of the Caribbean. In the United States, Florida and Hawaii have the highest incidences of HTLV-1-related diseases. Recent evidence also suggests HTLV-1 may be a re-emerging health threat in some global regions, such as Australia. The high-risk subtype HPVs are causally linked with cervical cancers and head-and-neck carcinomas which often have poor clinical outcomes and high mortality rates. Our research has demonstrated that the HTLV-1 and high-risk subtype HPVs encode proteins that cooperate with cellular oncogenes, including c-Myc, through molecular interactions with transcriptional coactivators and the differential modulation of p53-regulated pro-survival signals to promote cellular immortalization/transformation in vitro and tumorigenesis in in vivo xenograft models of HTLV-1-induced T-cell lymphoma and HPV-induced carcinomas. Importantly, these studies have revealed several key players which are essential for the survival of virus-infected tumor cells, as determined through siRNA-knockdown experiments, and may be candidates for the translational design of targeted therapeutics.
My laboratory’s research is supported by grant funding from the National Cancer Institute/National Institutes of Health.
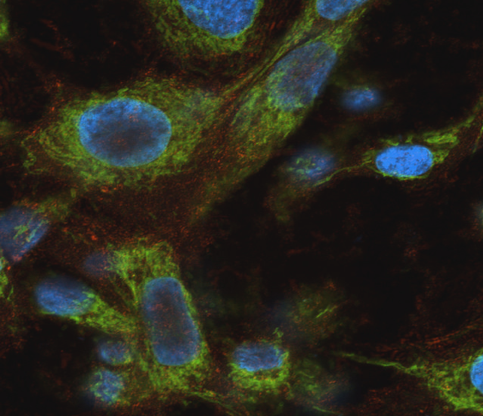
Photo: Mitochondrial DNA-damage induced by an HTLV-1 mutant provirus.
Selected Publications
Bowley T, Malu A, Adams NM, Savage J, Saberi M, VanderHagen M, Alame R, Keating M, Yates C, and R Harrod (2024). The HTLV-1 latency-maintenance factor p30II induces the phosphorylation and hypoxia-independent mitochondrial targeting of TIGAR analogous to tyrosine kinase receptor-signaling and suppresses oncogene-induced oxidative toxicity. Journal of Antivirals & Antiretrovirals, 16(2): 100000313 [1-16; open access].
Yapindi L, Bowley T, Kurtaneck N, Bergeson RL, James K, Wilbourne J, Harrod CK, Hernandez BY, Emerling BM, Yates C, and R Harrod (2023). Activation of p53-regulated pro-survival signals and hypoxia-independent mitochondrial targeting of TIGAR by human papillomavirus E6 oncoproteins. Virology 585:1-20.
Yapindi L, Hernandez BY, and R Harrod (2021) siRNA-Inhibition of TIGAR hypersensitizes human papillomavirus-transformed cells to apoptosis induced by chemotherapy drugs that cause oxidative stress. Journal of Antivirals & Antiretrovirals 13(4): 223 [open access].
Newman RA, Sastry KJ, Arav-Boger R, Matos R, and R Harrod (2020) Antiviral effects of oleandrin. Journal of Experimental Pharmacology 12: 503-515.
Harrod R (2019) Silencers of HTLV-1 and HTLV-2: the pX-encoded latency-maintenance factors. Retrovirology, in press [invited review].
Malu A, Hutchison T, Yapindi L, Smith K, Nelson K, Bergeson R, Pope J, Romeo M, Harrod C, Ratner L, Van Lint C, and R Harrod (2019) The human T-cell leukemia virus type-1 Tax oncoprotein dissociates NF-kappa-B p65RelA-Stathmin complexes and causes catastrophic mitotic spindle damage and genomic instability. Virology 535: 83-101.
Hutchison T, Yapindi L, Malu A, Newman RA, Sastry KJ, and R Harrod (2019) The botanical glycoside oleandrin inhibits HTLV-1 infectivity and Env-dependent virological synapse formation. Journal of Antivirals & Antiretrovirals, in press.
Hutchison T, Malu A, Yapindi L, Bergeson R, Peck K, Romeo M, Harrod C, Pope J, Smitherman L, Gwinn W, Ratner L, Yates C, and R Harrod (2018) The Tp53-Induced Glycolysis and Apoptosis Regulator mediates cooperation between HTLV-1 p30II and the retroviral oncoproteins Tax and HBZ and is highly expressed in an in vivo xenograft model of HTLV-1-induced lymphoma. Virology 520: 39-58.
Romeo M, Hutchison T, Malu A, White A, Kim J, Gardner R, Smith K, Nelson K, Bergeson R, McKee R, Harrod C, Ratner L, Lüscher B, Martinez E, and R Harrod (2018) The human T-cell leukemia virus type-1 p30II protein activates p53 and induces the TIGAR and suppresses oncogene-induced oxidative stress during viral carcinogenesis. Virology 518: 103-115.
Romeo MM, Ko B, Kim J, Brady R, Heatley HC, He J, Harrod CK, Barnett B, Ratner L, Lairmore MD, Martinez E, Lüscher B, Robson CN, Henriksson M, and R Harrod (2015). Acetylation of the c-MYC oncoprotein is required for cooperation with the HTLV-1 p30II accessory protein and the induction of oncogenic cellular transformation by p30II/c-MYC. Virology 476: 271-288.
R Harrod (2011) Inhibiting HDACs in a preclinical model of HTLV-1-induced Adult T-cell Lymphoma. Leukemia Res. 35: 1436-1437.
Sharma A, Awasthi S, Harrod CK, Matlock EF, Khan S, Xu L, Chan S, Yang H, Thammavaram CK, Rasor RA, Burns DK, Skiest DJ, Van Lint C, Girard AM, McGee M, Monnat RJ Jr, and R Harrod (2007) The Werner syndrome helicase is a cofactor for HIV-1 long terminal repeat transactivation and retroviral replication. J. Biol. Chem. 282: 12048-12057.
Nguyen TL-N, de Walque S, Veithen E, Deckoninck A, Martinelli V, de Launoit Y, Burny A, Harrod R, and C Van Lint (2007) Transcriptional regulation of the bovine leukemia virus promoter by the cyclic AMP response element modulator tau isoform. J. Biol. Chem. 282: 20854-20867.
Awasthi S, Sharma A, Wong K, Zhang J, Matlock EF, Rogers L, Motloch P, Takemoto S, Taguchi H, Cole MD, Lüscher B, Dittrich O, Tagami H, Nakatani Y, McGee M, Girard AM, Gaughan L, Robson CN, Monnat RJ Jr, and R Harrod (2005) An HTLV-1 enhancer of Myc transforming potential stabilizes Myc-TIP60 transcriptional interactions. Mol. Cell. Biol. 25: 6178-6198.
Nicot C, Harrod R, Ciminale V, and G Franchini (2005) Human T-cell leukemia/lymphoma virus type 1 nonstructural genes and their functions. Oncogene 24: 6026-6034.
Wong K, Sharma A, Awasthi S, Matlock EF, Rogers L, Van Lint C, Skiest DJ, Burns DK, and R Harrod (2005) HIV-1 Tat interactions with p300 and PCAF transcriptional coactivators inhibit histone acetylation and neurotrophin-signaling through CREB. J. Biol. Chem. 280: 9390-9399.
Wong K, Zhang J, Awasthi S, Sharma A, Rogers L, Matlock EF, Van Lint C, Karpova T, McNally J, and R Harrod (2004) Nerve growth factor receptor signaling induces histone acetyltransferase domain-dependent nuclear translocation of p300/CREB-binding protein-associated factor and hGCN5 acetyltransferases. J. Biol. Chem. 279: 55667-55674.
*Harrod R, Nacsa J, Van Lint C, Hansen J, Karpova T, McNally J, and G Franchini (2003) Human immunodeficiency virus type-1 Tat/co-activator acetyltransferase interactions inhibit p53K320-acetylation and tumor suppressor-responsive transcription. J. Biol. Chem. 278: 12310-12318 (*corresponding author).
Johnson JM, Harrod R, and G Franchini (2001) Molecular biology and pathogenesis of the HTLV-I. Int. J. Exp. Path. 82: 135-147.
Nicot C, and R Harrod (2000) Distinct p300-responsive mechanisms promote caspase-dependent apoptosis by HTLV-1 Tax. Mol. Cell. Biol. 20: 8580-8589.
Kuo YL, Tang Y, Harrod R, Cai P, and CZ Giam (2000) Kinase-inducible domain-like region of HTLV type 1 Tax is important for NF-kappaB activation. AIDS Research and Human Retroviruses 16: 1607-1612.
Harrod R, Kuo YL, Tang Y, Yao Y, Vassilev A, Nakatani Y, and CZ Giam (2000) p300 and P/CAF interact with HTLV-1 Tax in a multi-HAT/activator-enhancer complex. J. Biol. Chem. 275: 11852-11857.
Harrod R, Tang Y, Nicot C, Lu HS, Vassilev A, Nakatani Y, and CZ Giam (1998) An exposed KID-like domain in HTLV-1 Tax is responsible for the recruitment of co-activators CBP/p300. Mol. Cell. Biol. 18: 5052-5061.
Tang Y, Tie F, Boros I, Harrod R, Glover M, and CZ Giam (1998) An extended alpha-helix and specific amino acid residues opposite the DNA binding surface of the CREB basic domain are important for HTLV-1 Tax binding. J. Biol. Chem. 273: 27339-27346.