Richard S. Jones
Chair and Professor
Biological Sciences
| Office Location |
DLSB 333 |
| Phone |
214-768-3810 |
Education
Ph.D. Wesleyan UniversityPostdoc, Harvard University
Research Interests
Epigenetic Gene Regulation by Polycomb-Group Proteins
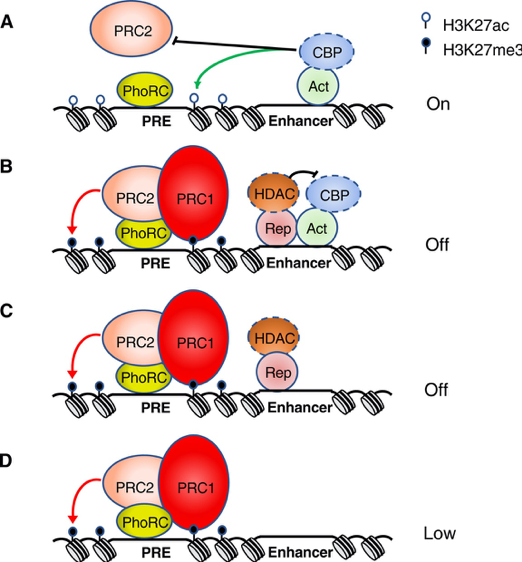
Model for regulation of PcG recruitment
The Jones lab studies the mechanisms by which a group of evolutionarily conserved epigenetic regulators, called the Polycomb-group (PcG), maintain the transcriptional silence of genes. PcG proteins play critical roles in regulating normal development in essentially all metazoans, functioning in largely similar ways in organisms ranging from mammals, insects, and even plants. The primary targets of PcG-mediated transcriptional repression are regulators of development and/or cell cycle progression. Among PcG targets are lineage-specific genes in embryonic and adult stem cells, and pluripotency genes when embryonic stem cells are induced to differentiate. Misexpression of human PcG proteins contributes to a wide range of cancers. Repression of pluripotency genes and the oncogenic effects of misregulated PcG proteins both involve de novo repression of target genes.
Our current focus is to define the molecular and biochemical mechanisms by which PcG-mediated gene silencing is initially established and to shed light on this important, but poorly understood, epigenetic phenomenon. In pursuit of this goal, the Jones lab uses a combination of genetic, immunological, biochemical and transgenic experimental approaches. Due to the high degree of conservation of PcG proteins, we are able to take advantage of the experimental tools available with the Drosophila melanogaster (fruit fly) model system with confidence that our findings will also provide insight into the mechanisms by which PcG proteins contribute to mammalian development and oncogenesis.
Ongoing projects include identifying proteins that are associated with a PcG target gene and dissecting their respective contributions to the establishment of transcriptional silencing.
Selected Publications
https://www.ncbi.nlm.nih.gov/myncbi/1DS2ez8Jk6kQ9/bibliography/public/
Ghotbi, E., P. Ye, T. Ervin, A. Kum, J. Benes, and R.S. Jones (2021) Polycomb-group recruitment to a Drosophila target gene is the default state that is inhibited by a transcriptional activator. Science Advances 7: eabg1556.
Ghotbi, E., K. Lackey, V. Wong, K.T. Thompson, E.G. Caston, M. Haddadi, J. Benes, and R.S. Jones (2020) Differential contributions of DNA binding proteins to Polycomb Response Element activity at the Drosophila giant gene. Genetics 214: 623-634.
AlHaj Abed, J., E. Ghotbi Ravandi, P. Ye, A. Frolov, J. Benes, and R.S. Jones (2018) De novo recruitment of Polycomb-group proteins. Development. In Revision.
AlHaj Abed, J., C.L. Cheng, C.R. Crowell, L.L. Madigan, E. Onwuegbuchu, S. Desai, J. Benes, and R.S. Jones (2013) Mapping Polycomb Response Elements at the Drosophila melanogaster giant locus. G3 3: 2297-2304.
AlHaj Abed, J. and R.S. Jones (2012) H3K36me3 key to Polycomb-mediated gene silencing in lineage specification. Nat. Struct. Mol. Biol. 19: 1214-1215.
Lee, N., H. Erdjument-Bromage, P. Tempst, R.S. Jones, and Y. Zhang (2009) The H3K4 demethylase Lid associates with and inhibits the histone deacetylase Rpd3. Mol. Cell. Biol. 29:1401-1410.
Joshi, P., E.A. Carrington, L. Wang, C.S. Ketel, E.L. Miller, R.S. Jones, and J.A. Simon (2008) Dominant alleles identify SET domain residues required for histone methyltransferase of Polycomb repressive complex 2. J. Biol. Chem. 283: 27757-27766.
Savla, U., J. Benes, J. Zhang, and R.S. Jones (2008) Recruitment of Drosophila Polycomb-group proteins by Polycomblike, a component of a novel protein complex in larvae. Development. 135: 813-817.
Jones, R.S. (2007) Reversing the irreversible. Nature 450: 357-359.
Lee, N., J. Zhang, R.J. Klose, H. Erdjument-Bromage, P. Tempst, R.S. Jones, and Y. Zhang (2007) The trithorax-group protein Lid is a histone H3 trimethyl-Lys4 demethylase. Nat. Struct. Mol. Biol. 14:341-343.
Wang, J., N. Jahren, M.L. Vargas, E.F. Andersen, J. Benes, J. Zhang, E.L. Miller, R.S. Jones, and J.A. Simon (2006) Alternative ESC and ESC-Like subunits of a Polycomb group histone methyltransferase complex are differentially deployed during Drosophila development. Mol. Cell. Biol. 26:2637-2647.
Wang, H., L. Wang, H. Erdjument-Bromage, M. Vidal, P. Tempst, R.S. Jones, and Y. Zhang (2004) Role of histone H2A ubiquitination in Polycomb silencing. Nature 431:873-878.
Wang, L., J.L. Brown, R. Cao, Y. Zhang, J.A. Kassis, and R.S. Jones (2004) Hierarchical recruitment of Polycomb-group silencing complexes. Mol. Cell 14:637-646.
Sedkov, Y., E. Cho, S. Petruk, L. Cherbas, S.T. Smith, R.S. Jones, P. Cherbas, E. Canaani, J.B. Jaynes, and A. Mazo (2003) Methylation at lysine 4 of histone H3 in ecdysone-dependent development of Drosophila. Nature 426: 78-83.
Cao, R., L. Wang, H. Wang, L. Xia, H. Erdjument-Bromage, P. Tempst, R.S. Jones, and Y. Zhang (2002) Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science 298:1039-1043.
O’Connell, S., L. Wang, S. Roberts, C.A. Jones, R. Saint, and R.S. Jones (2001) Polycomblike PHD fingers mediate conserved interaction with Enhancer of zeste protein. J. Biol. Chem 276: 43065-43073.